 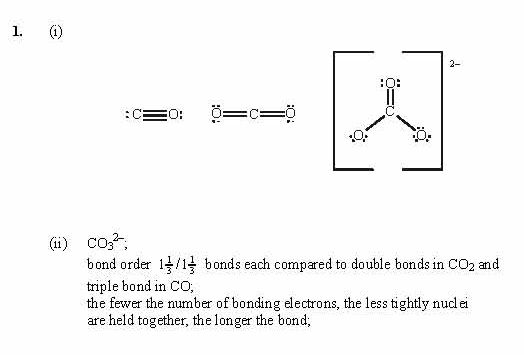
What is the formal charge of each atom in the CO molecule? 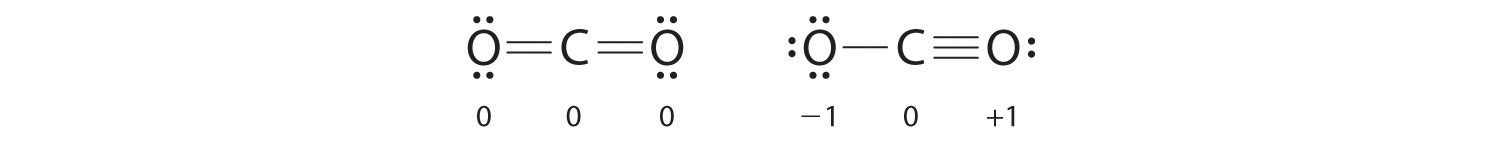
The Lewis structure of CH2O can be useful in understanding this reaction and predicting the products that may form in other chemical reactions involving CH2O. This is because CO can react with CH2O to form a stable product, and the amount of CO consumed can be measured to determine the amount of CH2O present. Analytical chemistry: CO is used in analytical chemistry to detect and measure the presence of various compounds, including CH2O.Fuel for combustion engines: CO is used as a fuel for combustion engines such as internal combustion engines and gas turbines.Chemical synthesis: CO is used as a building block for the synthesis of organic compounds such as methanol, acetic acid, and formaldehyde.Production of metal products: CO is used as a reducing agent in the production of iron, steel, and other metals.Applications of COĭespite its toxicity, CO has many important industrial applications. This property makes it useful in the production of metal and chemical products, as well as in the reduction of pollutants in the environment. As a result, the oxygen atom has a partial negative charge, while the carbon atom has a partial positive charge.ĬO is a strong reducing agent, meaning it donates electrons to other atoms or molecules. The bond between carbon and oxygen is a polar covalent bond, with oxygen being more electronegative than carbon. Reactivity: Highly reactive with oxygen and other oxidizing agentsĬO has a linear molecular geometry, with a bond angle of 180 degrees.CO is a byproduct of incomplete combustion, such as that found in car exhaust or gas stoves, and can build up in enclosed spaces without proper ventilation. It is a highly toxic gas that can be lethal in high concentrations. Properties of COĬarbon monoxide (CO) is a colorless, odorless, and tasteless gas at standard temperature and pressure. The triple bond between carbon and oxygen indicates that they share three pairs of electrons, fulfilling the octet rule for both atoms. The final Lewis Structure of CO should look like this: In CO, the formal charge of Carbon (C) is 0, and the formal charge of Oxygen (O) is -1. The formal charge of an atom is the difference between its valence electrons and the number of electrons it has in the Lewis Structure. In CO, both Carbon (C) and Oxygen (O) have a full octet.Ĭalculate the formal charge for each atom in the molecule. Place any remaining valence electrons around the central atom, Carbon (C).Ĭheck to make sure that all atoms in the molecule have a full octet (except for Hydrogen (H), which only needs 2 electrons to fill its outer shell). Place these electrons around the Oxygen (O) atom, giving it a full octet (8 electrons). In CO, we have 4 valence electrons left after the single bond between C and O. Place the remaining valence electrons around the atoms in the molecule. Distribute the remaining valence electrons. Connect Carbon (C) and Oxygen (O) atoms with a single bond.Ĥ. In a Lewis Structure, a single bond is represented by a straight line (-) between the two atoms. In CO, Carbon (C) is the central atom because it has the lower electronegativity of the two atoms. The central atom in a Lewis Structure is usually the atom with the lowest electronegativity. 
Therefore, the total number of valence electrons in CO is 10. Carbon (C) has 4 valence electrons, and Oxygen (O) has 6 valence electrons. To determine the total number of valence electrons in CO, you need to add up the valence electrons of each atom in the molecule. Determine the total number of valence electrons in CO. How to Draw the Lewis Structure for COĭrawing the CO Lewis structure involves several steps:ġ. In this article, we will explore how to draw the Lewis structure for CO, and the significance of CO in various fields of chemistry. The Lewis structure for CO (carbon monoxide) is a crucial component of understanding its molecular properties and reactivity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
